Prof Matthew Costa
(Chief Investigator)
Dr Susan Wagland
(Trial Manager)

- To quantify and draw inferences on observed differences in Achilles Tendon Rupture Score between the trial treatment groups at 9 months after injury.
- To quantify and draw inferences on observed differences in Achilles Tendon Rupture Score between the trial treatment groups at 8 weeks, 3 and 6 months after the injury.
- To identify any differences in health-related quality of life between the trial treatment groups in the first 9 months after the injury.
- To determine the complication rate between the trial treatment groups in the first 9 months after the injury.
- To investigate, using appropriate statistical and economic analytical methods, the resource use, costs and comparative cost effectiveness between the trial treatment groups.
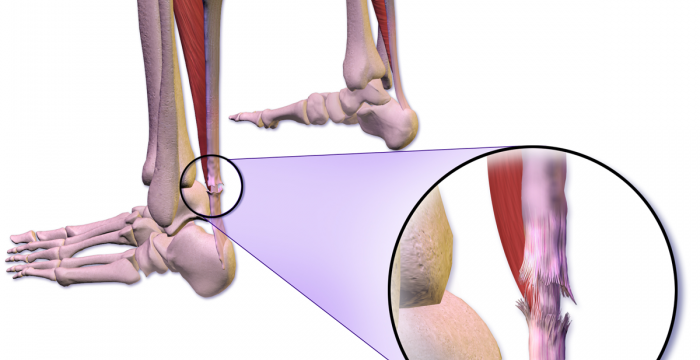
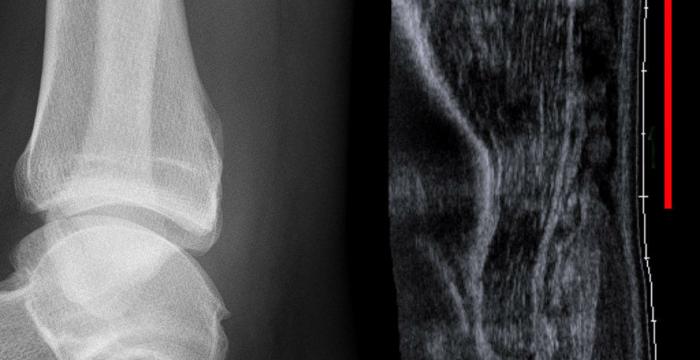
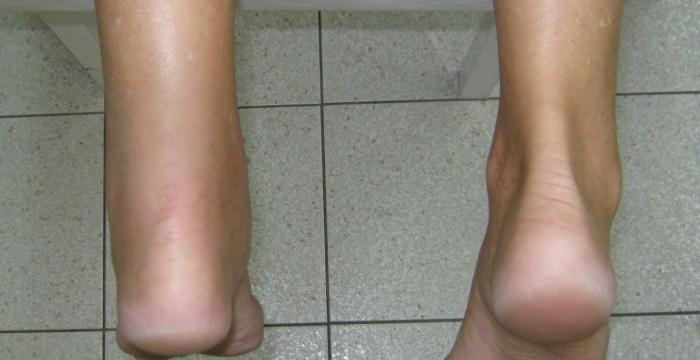
The following outcome measures will be collected:
- Achilles Tendon Rupture Score
- EQ-5D-5L
- Resource use
- Complications

NEWS
UKSTAR's Statistical Analysis Plan is published in the journal Trials. See it here. Cast versus functional brace in the rehabilitation of patients with a rupture of the Achilles tendon: statistical analysis plan for the UK study of tendo Achilles rehabilitation (UK STAR) multi-centre randomised controlled trial
UKSTAR has completed data collection. The last follow-up (the definition of End of Study in the Protocol) took place on Tuesday 12 March. We received 504 responses from participants at the 9 month timepoint, the trial's primary outcome - which represents a 93% response rate, unusually high in this kind of trial. A MASSIVE THANKS to all our participants.
UKSTAR is now CLOSED to RECRUITMENT. Please continue to send in your follow-up questionnaires at 8 weeks, 3 months, 6 months and 9 months after entry into the trial.
31st May 2018: UKSTAR closes to recruitment with a massive 540 participants (2 less than the number on the thermometer view above). Well done everyone!
16th January 2018: Aberdeen recruited the 400th patient to UKSTAR. Congratulations to the team.
On 29th June 2017 Aberdeen recruited to the UKSTAR our 200th patient. Congratulations to the team.
Congratulations to Leeds for becoming our third highest recruiting site in the country.
First married couple in the UKSTAR trial recruited by Aberdeen team.
Congratulations to Dundee for being the UKSTAR 2nd highest recruiting site.
Congratulations to the Aberdeen trial team for recruiting the largest number of participants into the UKSTAR trial to date.
Congratulations to King's College Hospital for recruiting the FIRST participant into the UKSTAR trial.
CONNECTIONS
UKSTAR's protocol is published in BMJ Open.
Read more about the UKSTAR Trial on the Oxford Trauma website.
See the UKSTAR Trial on ISRCTN - primary clinical trial registry
See the UKSTAR Trial on Oxford University Hospitals NHS Foundation Trust website
British Orthopaedic Association: For more reading about Trauma and Orthopaedics in the UK.
Access the article about the trial in the NIHR Journals Library